Flagship Project
A computational detector for tear-level hemodynamics in chronic aortic dissection.
This project develops a novel computational method that localizes and quantifies hemodynamically active true-lumen and false-lumen communication sites from time-resolved 4D Flow MRI, producing structured tear-level outputs that can support future cohort-scale interpretation.
Peak orthogonal flow representation
Representative peak orthogonal-flow signal from a chronic dissection case. The detector operates on this distributed hemodynamic input.
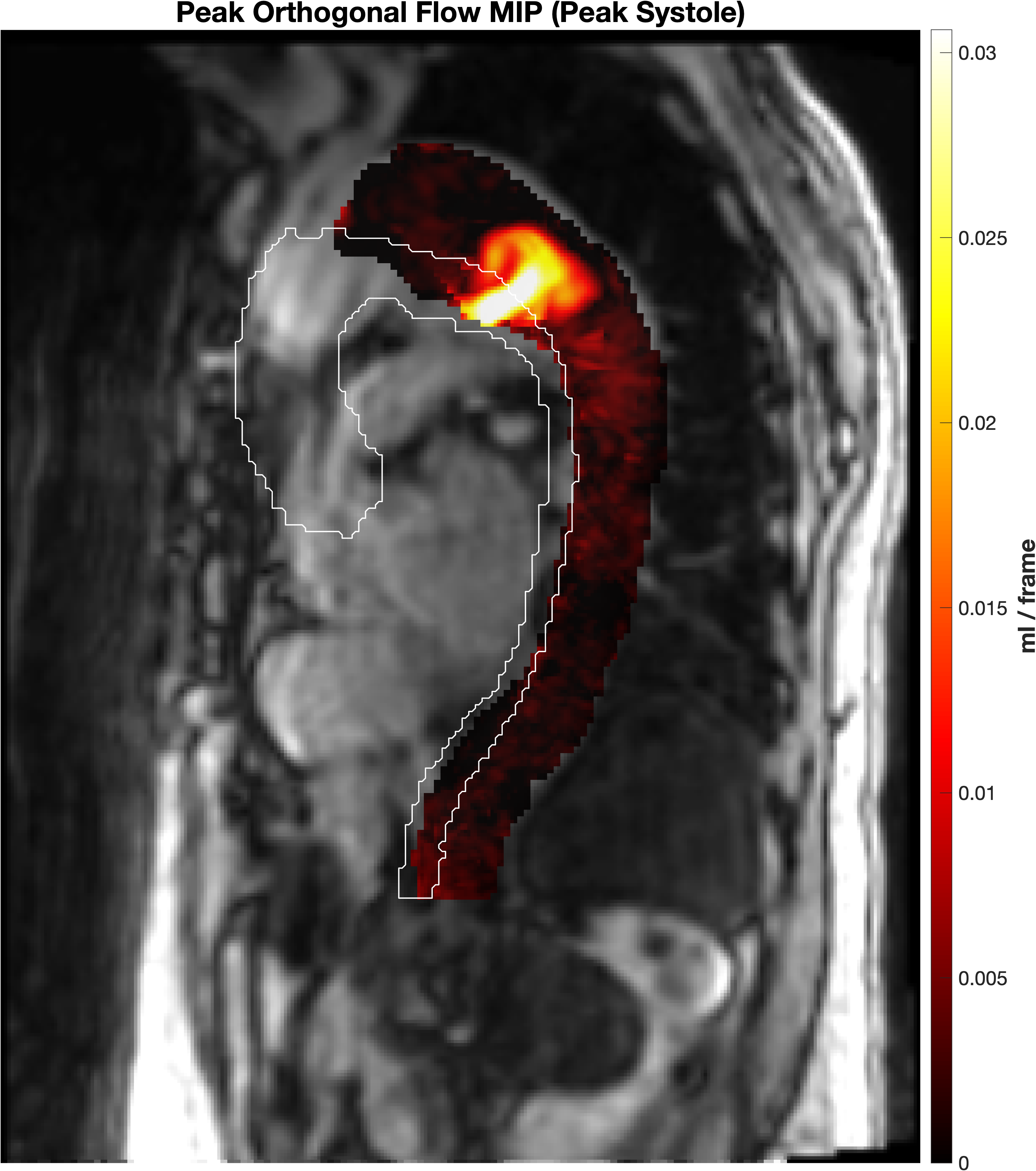
The method converts this signal into discrete tear-level measurements.
Project Snapshot
The project links a clinically important disease mechanism, a technically difficult MRI modality, a detector-based computational method, and a future interpretation pathway.
- Health problem
- Persistent TL/FL communication in chronic dissection
- Data modality
- 4D Flow MRI with time-resolved 3D velocity fields
- Computational contribution
- Automated tear localization, sizing, and flow quantification
- Future trajectory
- From tear outputs to agentic patient interpretation
Clinical Problem
Persistent TL/FL communication is a mechanistic problem that anatomy alone does not resolve
In chronic aortic dissection, the false lumen often remains perfused through multiple tears or fenestrations connecting it to the true lumen. Those communication sites matter because they sustain patient-specific flow exchange that may contribute to persistent false-lumen pressurization, adverse remodeling, and downstream intervention risk.
Aortic dissection is a life-threatening condition in which an intimal tear creates a false lumen within the aortic wall.
In chronic disease, what often matters is not anatomy alone but continued communication between true and false lumens.
That makes tear-site hemodynamics a plausible mechanistic target for measurement rather than a purely descriptive imaging finding.
Clinical overview
True lumen, false lumen, and entry-tear anatomy
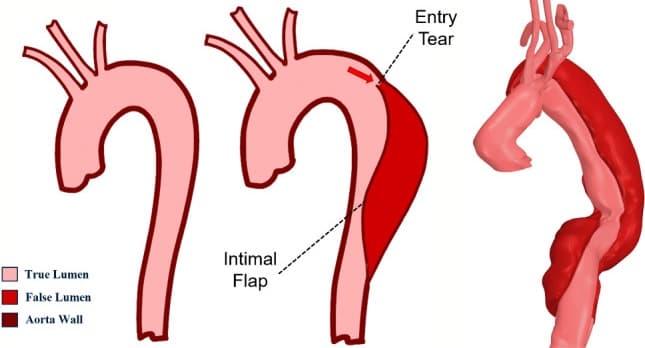
Illustrations of normal and dissected aortic anatomy with a patient-specific dissected aorta, highlighting true lumen, false lumen, and entry-tear anatomy. Adapted from Marimuthu et al., Biomechanics and Modeling in Mechanobiology, 2025.
Why This Is Hard
4D Flow MRI makes the relevant physiology visible, but not easy to measure
The computational problem arises because the clinically relevant signal is distributed across space, direction, and time within a complex dissected geometry.
High-dimensional data
4D Flow MRI is volumetric, time-resolved, and vector-valued, so the clinically relevant signal is distributed across space, direction, and the cardiac cycle.
Complex geometry
Dissection anatomy creates narrow, irregular interfaces between true and false lumens, making conventional plane-based analysis cumbersome and incomplete.
Interpretability matters
The method must return clinically meaningful communication sites and flow descriptors, not just a visually interesting map without structured outputs.
Novelty in one sentence
This is not an imaging workflow that stops at visualization. It is a detector that produces standardized tear-level measurements from 4D Flow MRI.
Computational Contribution
The detector formalizes tear-site analysis as a computational problem
The key contribution is not simply a new visualization. It is a detector that formalizes tear-site analysis as a computational problem, identifies discrete communication sites, and reports standardized tear-level descriptors that can be compared across patients and carried into downstream models.
Problem formulation
Recast dissection analysis around hemodynamically active TL/FL communication sites rather than anatomy alone.
Algorithmic strategy
Use orthogonal-flow projections at the dissection interface to turn a difficult velocity field into localized candidate tears.
Structured output
Produce tear-level objects with location, size, and exchange readouts that can support later interpretation and modeling.
Detector Outputs
The main result is a set of structured tear-level measurements
The output is not a single global score. It is a set of tear-level measurements that preserve location, geometry, and exchange behavior, making the result both interpretable to humans and useful for downstream computation.
Detector output
Localized tear plus site-specific flow curve
Example output showing how the method turns a distributed flow field into a discrete candidate site with associated temporal exchange behavior.

This figure is the core translational product of the method: a localized communication site with structured measurements that can be reviewed, compared, and modeled.
Output structure
- Localized tear candidates embedded in patient-specific anatomy
- Estimated tear size and spatial extent
- Flow-time curves and directional exchange readouts for each site
- Structured tear-level features that can be aggregated across patients
Localized exchange is not uniformly distributed across the dissected aorta. That heterogeneity is one reason a tear-level detector is more informative than a global impression of the flow field.
Method
How the detector gets there
The detector projects each voxel's velocity vector onto the local orthogonal direction between false-lumen and true-lumen centerlines, restricts analysis to the dissection interface, identifies clustered exchange hotspots, and reports tear-level geometry and flow readouts for each detected site.
Pipeline
Detector overview
Orthogonal-flow representation, interface restriction, and tear-level quantification.
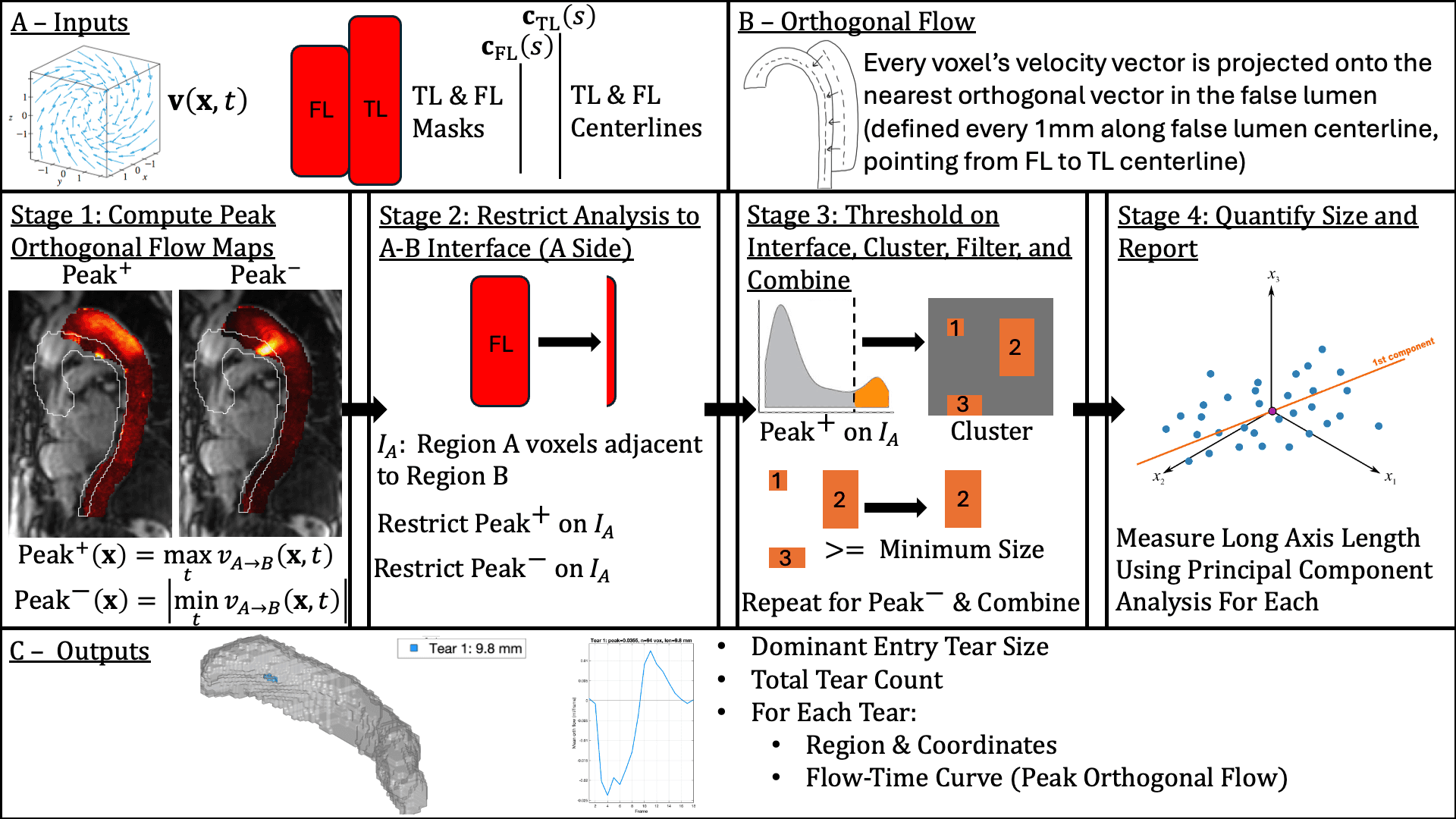
The method is designed to move from high-dimensional velocity data to reproducible tear-level outputs.
Represent localized exchange
Convert the raw velocity field into orthogonal-flow maps that explicitly quantify TL-to-FL and FL-to-TL communication.
Restrict the search space
Constrain analysis to voxels adjacent to the dissection interface where tears are physiologically plausible, improving specificity.
Detect and quantify
Cluster candidate regions, remove small artifacts, and report tear-level geometry and flow descriptors such as size, peak orthogonal flow, and net exchange.
Why These Outputs Matter
Localized exchange measurements can carry clinically meaningful signal
These supporting figures illustrate why localized tear-level outputs are worth keeping: they connect with established hemodynamics, show selective outcome signal, and support quantitative size estimation.
Clinical relevance
Connection to established hemodynamics
The most active regional peak orthogonal flow tracks with false-lumen ejection fraction, linking localized measurements to broader hemodynamic behavior.
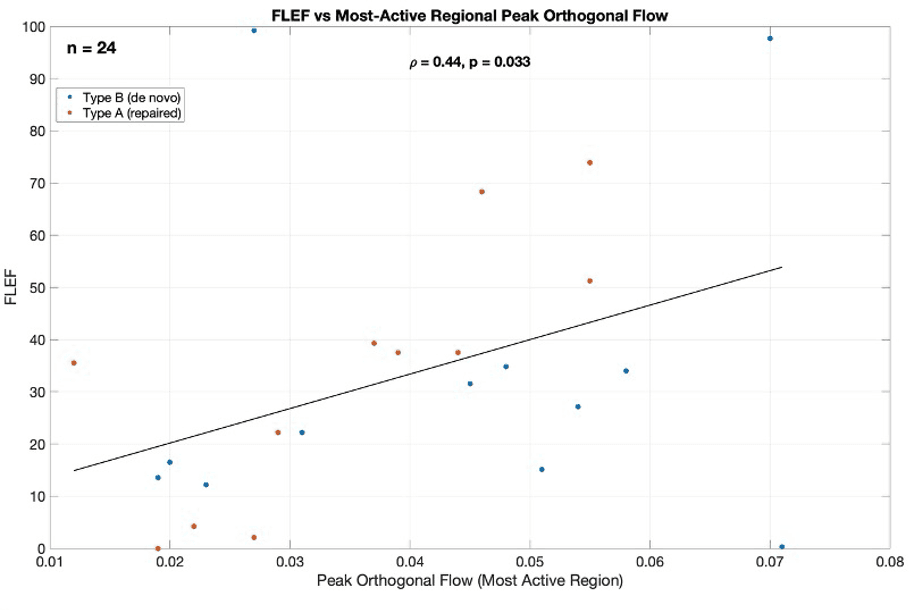
This helps position tear-level exchange as clinically relevant rather than merely descriptive.
Clinical relevance
Selective signal for outcomes
Peak orthogonal flow shows separation by outcome where other summarized metrics appear less informative.
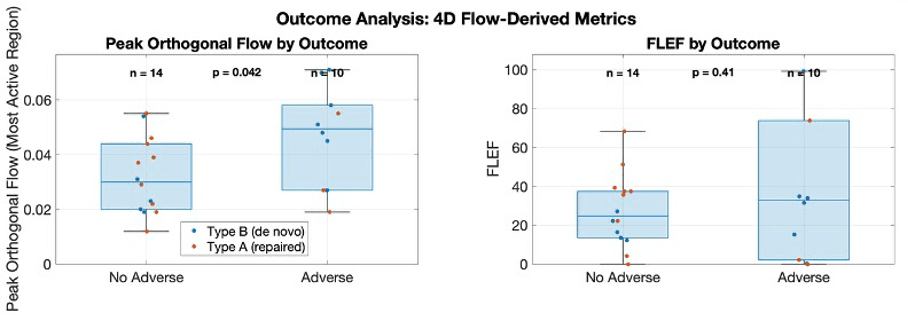
The implication is not that one figure proves prognosis, but that localized exchange severity may carry signal worth following rigorously.
Measurement validation
Quantitative size estimation matters too
A detector becomes more credible when it can be evaluated not only on whether it highlights a region, but also on whether it produces measurable quantities that can be compared against anatomic reference data.
Measurement validation
Quantitative size estimation
Detector-derived tear sizing can be compared against anatomic reference measurements rather than being judged qualitatively.
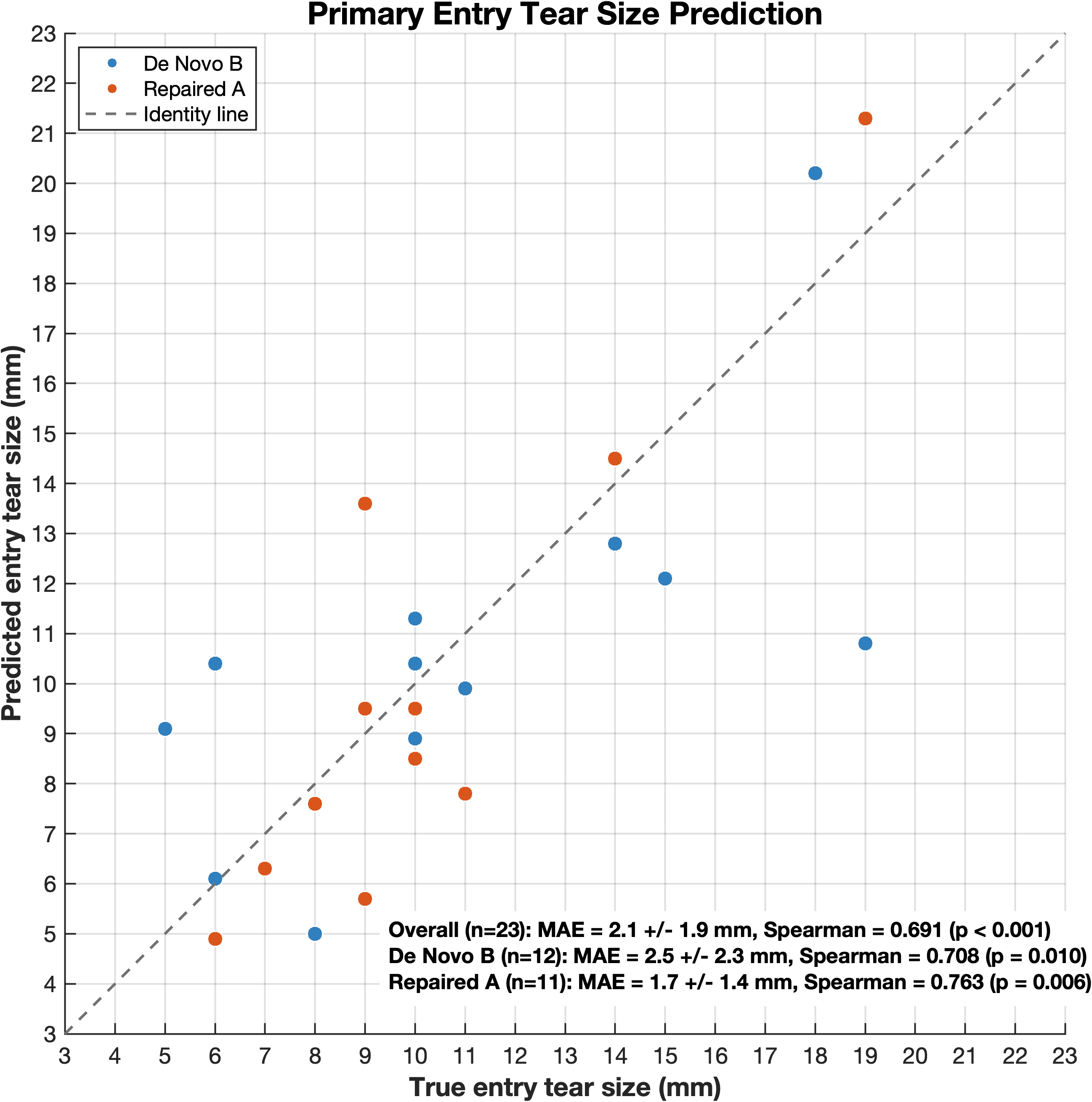
Size estimation strengthens the method as a real measurement pipeline, not only a hotspot detector.
Evaluation
Validation spans controlled ground truth and retrospective translation
The validation logic intentionally spans controlled ground truth and retrospective human data. That combination matters because the method is trying to do two things at once: detect real communication sites and produce clinically meaningful measurements.
Detector fidelity
Evaluate whether the algorithm localizes discrete communication sites and produces stable, interpretable tear-level descriptors.
Phantom benchmarking
Use MRI-compatible pulsatile flow experiments and patient-specific 3D-printed phantoms to test localization and size accuracy against controlled ground truth.
Retrospective cohort translation
Compare detector outputs against high-resolution anatomic reference data and study whether tear-level features carry clinically relevant signal across patient cohorts.
Supporting validation
Phantom validation
This phantom workflow is included as a supporting validation pathway only. It is distinct from retrospective cohort evaluation and should not be interpreted as the main clinical story of the project.
The phantom workflow provides controlled ground truth for site location, size, and exchange-related measurements under MRI-compatible pulsatile flow conditions.
Patient-specific phantom workflow
Segmentation, post-processing, and 3D printing for controlled comparison against known phantom ground truth.

Limitations
Current boundaries of the method
- Detection depends on the quality of lumen masks, centerlines, and 4D Flow acquisition.
- 4D Flow MRI trades anatomic resolution for rich hemodynamic information, so very small communication sites may remain difficult to resolve reliably.
- Current outputs quantify exchange at detected sites, but they do not by themselves replace longitudinal clinical judgment or anatomy-based review.
Future Direction
From tear detection to agentic interpretation
Each detected tear can serve as a structured unit of evidence. Those tear-level agents can then be aggregated with anatomy and clinical variables to support future cohort-scale interpretation and patient-level risk scoring.
Multi-agent patient risk model
Tear-level agents
Tear agent 1
Tear agent 2
Tear agent n
Anatomic features
Multi-agent fusion
Patient-level coordinator
Aggregates all detected tears in a patient and fuses tear-level outputs with anatomic and clinical context.
Clinical features
Patient-specific risk score